Understanding Cerebral Hemorrhage: Definition and Classification
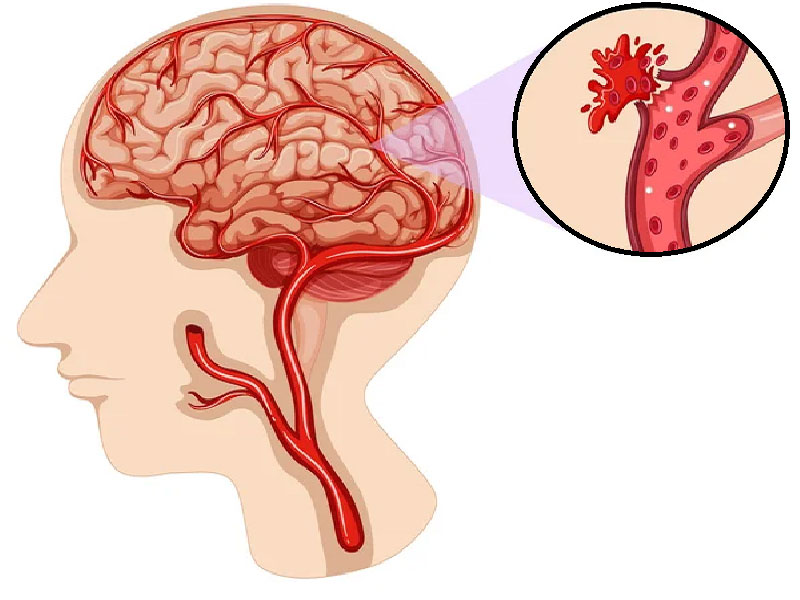
A cerebral hemorrhage represents a life-threatening neurological emergency. Blood spills into brain tissue when arterial vessels rupture. This isn't simply a bleeding episode, it's a cascade of physiological disruption that demands immediate intervention. The brain, unlike other organs, has virtually no capacity for additional blood volume; even modest accumulation triggers dangerous pressure buildup.
Clinically, hemorrhages divide into two broad categories based on mechanism. Traumatic bleeding follows head injury with considerable force, often from motor accidents, falls, or assaults; these are largely preventable through protective measures. Spontaneous hemorrhages arise without external trauma, stemming instead from underlying vascular pathology or systemic disease, hypertension, anticoagulation therapy, or structural vessel abnormalities. The distinction matters enormously for prevention strategy and long-term prognosis.
Not all bleeds present identically. Some patients experience gradual symptom onset over hours or even days (subdural hematomas in elderly patients, for instance), while others deteriorate within minutes. This temporal variability complicates both patient recognition and medical response timing.
The Anatomical Spectrum: Where Brain Bleeding Occurs
Location determines everything in cerebral hemorrhage. The brain is surrounded by three membrane layers, the dura mater (outermost), arachnoid, and pia mater (innermost), plus cerebrospinal fluid. Blood can accumulate in different spaces, each producing distinct clinical consequences.
Extradural hematoma develops between the skull bone and dura mater, typically after high-impact trauma causing skull fractures. Middle meningeal artery rupture is the classical culprit. Patients often describe a lucid interval: initial unconsciousness, then apparent recovery, followed by sudden deterioration. Without urgent surgical drainage, this pattern carries mortality rates approaching 50% in untreated cases.
Subdural hematoma accumulates in the space between dura and brain tissue itself. Veins crossing this compartment tear from sudden acceleration-deceleration forces. Elderly patients on anticoagulants can develop subdural bleeding from minor head bumps that younger people would dismiss, the fragile cerebral bridging veins rupture with minimal trauma. Chronic subdural hematomas, which develop slowly over weeks, mimic dementia: confusion, gait disturbance, personality changes. Acute subdural bleeds in younger patients (usually from severe head injury) present more dramatically.
Intraparenchymal hemorrhage (bleeding directly within brain tissue) typically results from chronic hypertension rupturing small penetrating vessels. The putamen and thalamus regions account for roughly 50% of cases; cerebellar and brainstem hemorrhages, though less common, carry worse prognosis. Volume matters critically, bleeds exceeding 30 cubic centimeters generally require surgical intervention; smaller volumes may be managed medically if the patient remains stable.
Subarachnoid hemorrhage (SAH) involves the space containing cerebrospinal fluid between arachnoid and pia. Ruptured cerebral aneurysms account for approximately 80-90% of non-traumatic SAH cases. The sudden release of blood into this space triggers vasospasm, sustained artery constriction lasting days or weeks, which paradoxically causes delayed stroke despite hemorrhage control. Patients report the worst headache of their life, often accompanied by neck stiffness, photophobia, and nausea.
Intraventricular hemorrhage occurs when blood enters the fluid-filled cavities deep within the brain. Extension into ventricles frequently complicates other hemorrhage types and substantially worsens outcomes through obstructive hydrocephalus (fluid backup causing increased intracranial pressure).
What Causes Brain Bleeding: Primary Risk Factors
Uncontrolled hypertension remains the leading preventable cause of spontaneous cerebral hemorrhage, responsible for approximately 50-60% of cases in most populations. Chronic high blood pressure remodels vessel walls through a process called lipohyalinosis, creating microaneurysms (Charcot-Bouchard aneurysms) prone to rupture. Interestingly, the relationship isn't linear, acute pressure spikes matter less than cumulative, long-standing elevation.
Head trauma severity varies enormously. Mild traumatic brain injury may never produce bleeding; moderate to severe injuries routinely do. Sports-related concussions occasionally progress to delayed hematomas, particularly in athletes who resume activity too quickly.
Arteriovenous malformations (AVMs) represent abnormal tangle-like connections between arteries and veins, bypassing normal capillary networks. These vessels have structurally weak walls. Annual rupture risk ranges from 1% to 4%, but prior hemorrhage increases future risk significantly. Many AVMs are discovered incidentally during imaging for unrelated symptoms.
Cerebral aneurysms, focal outpouchings of arterial walls, occur in roughly 2-3% of autopsy series, yet only a fraction rupture during life. Size and location predict risk: aneurysms larger than 7 millimeters carry higher rupture potential, as do those in the anterior communicating artery compared to other sites. Family history of aneurysm or polycystic kidney disease elevates personal risk substantially.
Brain tumors seldom cause hemorrhage directly but destabilize adjacent vessels or erode into vascular structures. Glioblastomas and metastases demonstrate particularly high hemorrhage propensity. Routine medical screening doesn't prevent this complication, but awareness of symptoms prompts earlier diagnosis.
Anticoagulation therapy (warfarin, apixaban, rivaroxaban) carries real bleeding risk in poorly controlled patients or those with underlying structural abnormalities. Supratherapeutic INR levels correlate with intracranial hemorrhage incidence. Antiplatelet agents (aspirin, clopidogrel) increase risk modestly but rarely cause catastrophic bleeding alone.
DIC (disseminated intravascular coagulation) represents systemic coagulation cascade activation from sepsis, trauma, malignancy, or obstetric complications. Paradoxically, widespread clot formation exhausts platelets and clotting factors, producing severe bleeding across multiple organs including the brain. Mortality exceeds 40% even with aggressive intervention.
Recognizing Brain Hemorrhage: Clinical Presentation and Symptom Progression
Symptom onset distinguishes hemorrhagic events from other neurological emergencies. Most patients describe sudden symptom appearance during activity, while walking, eating, or talking. This abruptness differs from stroke's sometimes gradual worsening and contrasts sharply with tumor symptoms developing over weeks or months.
The classic triad includes severe headache (often occipital or generalized), focal neurological deficits, and altered consciousness. Not every patient exhibits all three; presentation depends on hemorrhage location and size. A small frontal hemorrhage might produce subtle personality changes and mild headache, while a 40-milliliter cerebellar bleed causes sudden coma within hours.
Headache usually dominates initial presentation. Patients often use superlatives: "the worst headache of my life," "like an explosion in my head," "thunderclap onset." This quality differs from migraine's gradual build or tension headache's dull ache. Vomiting frequently accompanies severe cases, triggered by increased intracranial pressure stimulating the vomiting center. Some patients report photophobia and neck pain, symptoms mimicking meningitis.
Motor and sensory deficits appear contralateral to the bleed due to brain's crossing neural pathways. Weakness or numbness affecting one side, face, arm, leg, suggests hemispheric hemorrhage. Speech disturbance (dysarthria, slurred speech) versus language comprehension problems (aphasia, word-finding difficulty) localizes to different hemisphere regions: dysarthria implies cerebellar or brainstem involvement, aphasia indicates dominant (usually left) hemisphere frontal or temporal location. These distinctions guide emergency teams toward appropriate imaging.
Loss of consciousness ranges from mild drowsiness to deep coma unresponsive to pain. Rapid deterioration within hours of symptom onset typically indicates hematoma expansion or cerebral edema development. Seizures occur in 5-15% of acute hemorrhage cases, more common with lobar (cortical) bleeding. Some seizures represent initial presentation; others emerge during hospitalization as edema or inflammation peaks.
Complications arise as intracranial pressure climbs. Respiratory irregularities emerge, Cheyne-Stokes breathing (periodic crescendo-decrescendo pattern), central hyperventilation, or sudden apnea. Blood pressure becomes labile, sometimes critically elevated as the brain's autoregulatory mechanisms fail. Fever frequently develops, worsening neurological prognosis; elevated body temperature increases metabolic demands on already-damaged tissue.
Cerebral edema, swelling around the hemorrhage, develops over hours to days. This secondary injury often causes more damage than the initial bleed itself. Large hematomas expanding into deep structures risk herniation: the brain tissue physically shifts, compressing vital brainstem structures controlling consciousness and breathing. Herniation remains a leading cause of death in untreated severe hemorrhage.
Diagnostic Approach: Confirming and Characterizing Brain Bleeding
Clinical suspicion alone can't confirm hemorrhage. Imaging is mandatory. Non-contrast CT scan remains the emergency standard, fast, sensitive for acute bleeding, and available in virtually all hospitals. Blood appears white (hyperdense) against gray brain tissue. This imaging reveals hemorrhage location, volume (rough estimates possible), and mass effect (midline shift, ventricular compression). Multiple sequence capability, including multiplanar reconstruction, helps quantify exact volumes for prognostic scoring.
MRI provides superior soft tissue detail and detects underlying lesions causing bleeding: tumors, AVMs, cavernomas. However, MRI requires 30-60 minutes, unsuitable for unstable patients requiring immediate intervention. MRI excels in subacute phases (days 3-14 post-hemorrhage) when CT sensitivity wanes and patients are stable enough for longer scans.
CT angiography (CTA) identifies arterial aneurysms in subarachnoid hemorrhage with nearly 100% sensitivity; digital subtraction angiography follows for patients with SAH and negative CTA, capturing 15-20% of additional aneurysms missed on initial imaging. Angiography also evaluates vasospasm development around day 4-5.
Laboratory studies complement imaging. Coagulation studies (PT/INR, aPTT, platelet count, fibrinogen) identify patients with bleeding diathesis requiring correction. Elevated D-dimer and low fibrinogen suggest DIC. Troponin and BNP often rise after major hemorrhage due to catecholamine surge, though these don't require specific treatment, this "neurogenic stress cardiomyopathy" usually reverses spontaneously.
Lumbar puncture plays limited modern role since CT excludes hemorrhage definitively. SAH patients sometimes undergo LP if CT is negative but clinical suspicion remains high (xanthochromia, yellow cerebrospinal fluid discoloration, indicates old blood, confirming subarachnoid bleeding).
Medical Management: Initial Stabilization and Pressure Control
Once hemorrhage is confirmed, goals shift rapidly: prevent hematoma expansion, control intracranial pressure, and preserve neurological function. Initial management happens in ICU or stroke unit settings with continuous monitoring of vital signs, oxygen saturation, and neurological status via serial Glasgow Coma Scale assessments.
Blood pressure management presents paradox. Elevated pressure worsens bleeding and brain edema, yet hypotension reduces cerebral perfusion in injured tissue. Current guidelines recommend systolic targets between 140-180 mmHg in acute phases using titratable agents (nicardipine, labetalol, esmolol). Excessive lowering, below 140 mmHg, correlates with worse outcomes in some studies.
Reversal of anticoagulation is urgent in bleeding patients taking warfarin or novel anticoagulants. Warfarin reversal requires fresh frozen plasma or prothrombin complex concentrate plus vitamin K (though K takes 12-24 hours for effect). Direct factor Xa inhibitors (apixaban, rivaroxaban) and direct thrombin inhibitors (dabigatran) have specific reversal agents: andexanet alfa and idarucizumab respectively. Timely reversal can halt hematoma expansion in 20-30% of cases.
Intracranial pressure reduction employs multiple strategies. Head-of-bed elevation to 30 degrees improves venous drainage. Osmotic therapy, hypertonic saline (3% or 23.4%) or mannitol, draws fluid from brain tissue into vascular space, reducing edema. Hypertonic saline, preferred increasingly over mannitol, reduces ICP within minutes; effects last 4-8 hours. Sedation and paralysis (if intubated) reduce metabolism and ICP elevation from agitation. Hypothermia (32-35°C) shows promise in animal models but randomized human trials have not confirmed benefit; moderate hypothermia remains investigational.
Seizure prophylaxis uses levetiracetam (1-2 grams daily) rather than phenytoin, which impairs neurological assessment and may worsen long-term outcomes. Prophylaxis prevents early seizures (first 7 days) in lobar hemorrhages with high seizure risk; randomized data don't support extending beyond this window.
Fever management demands aggressive treatment. Elevated core temperature worsens brain injury; maintaining normothermia (<37°C) is standard. Acetaminophen, cooling blankets, and sedation adjustment all contribute.
Surgical Intervention: When and Why Operating Matters
Not all hemorrhages require surgery, patient age, hematoma size, location, and neurological grade determine candidacy. Generally, hematomas exceeding 30 milliliters with mass effect (midline shift >5mm, ventricular compression) warrant consideration. Smaller bleeds, even if symptomatic, often respond to medical management alone unless patient deteriorates.
Surgical options vary by location. Lobar (cortical) hematomas accessible near the brain surface undergo open craniotomy for clot evacuation, allowing inspection for underlying structural abnormalities (AVM, tumor, aneurysm). Incision size and brain retraction are minimized using neuronavigation and endoscopes when feasible. Deep basal ganglia or thalamic bleeds present greater technical challenge; some centers use stereotactic aspiration (needle-based catheter drainage under imaging guidance) or neuroendoscopy, though these approaches haven't definitively outperformed craniotomy in randomized trials.
Cerebellar hemorrhage requires special urgency. Even modest volumes (>5 milliliters) can compress the fourth ventricle, obstructing cerebrospinal fluid flow and causing sudden coma or death within hours. Most neurosurgeons recommend surgical drainage for cerebellar hematomas >3 centimeters or those with ventricular involvement, regardless of patient age or initial neurological grade.
Subarachnoid hemorrhage with aneurysm demands aneurysm repair, either microsurgical clipping or endovascular coil embolization, as soon as technically feasible. Early repair (within 72 hours) allows aggressive blood pressure elevation and hyperdynamic therapy to prevent delayed ischemia from vasospasm. Coil embolization has become predominant due to lower rebleeding rates and less invasiveness, though some aneurysm morphologies still require open surgery.
Hematoma evacuation timing remains debated. Animal data suggest benefit from evacuation within 12 hours, but human randomized trials (STITCH Cr trial) showed no mortality benefit from early surgery versus conservative management. Neurological deterioration or imaging evidence of hematoma expansion remains the strongest indication for urgent intervention.
Intracerebral hemorrhage outcomes depend partly on surgical volume; high-volume centers (>50 ICH cases annually) report better functional outcomes than low-volume centers. Specialized neurological care facilities with dedicated neurosurgical teams and ICUs optimize perioperative management.
Rehabilitation and Recovery: The Post-Acute Phase
Survival isn't the endpoint, functional recovery determines quality of life. Brain tissue, unlike liver or muscle, regenerates minimally. Rehabilitation exploits neuroplasticity: surviving neurons rewire connections, compensating for lost function. This process peaks in the first 3-6 months but continues for years.
Physical therapy begins immediately once patients stabilize, even passive range-of-motion exercises prevent contractures in paralyzed limbs. Active therapy escalates as consciousness improves: sitting balance, standing, ambulation retraining. Robotics-assisted devices show promise in some studies, allowing high-repetition practice without therapist fatigue. Walking-focused training produces better ambulatory outcomes than arm-centric therapy alone.
Speech and language therapy addresses dysarthria (slurred speech mechanics) versus aphasia (language comprehension or production deficits). Aphasia recovery depends on lesion location and extent; some patients regain substantial function over months, while others plateau. Early intensive speech therapy (5+ sessions weekly) correlates with better outcomes than intermittent approaches.
Cognitive rehabilitation targets attention, memory, executive function disturbances. Frontal or temporal hemorrhages frequently cause these deficits; patients may be physically recovered yet unable to work due to concentration or judgment impairment. Computer-based cognitive training shows modest benefits; real-world task practice (cooking, money management, job simulation) transfers better to daily functioning.
Depression and anxiety affect 30-40% of stroke survivors, including hemorrhage patients. Antidepressants (SSRIs) improve mood and may enhance rehabilitation participation. Psychotherapy and peer support groups address emotional aspects of disability.
Return-to-work timelines vary dramatically. Simple sedentary jobs may be feasible within weeks post-hemorrhage; complex safety-sensitive roles (aviation, surgery, commercial driving) require clearance, sometimes permanently denied after major deficits. Vocational rehabilitation specialists assess capabilities objectively.
Long-term outcomes: 30-day mortality ranges from 20% (small lobar bleeds) to 50%+ (large basal ganglia or brainstem hemorrhages). Of survivors, roughly 50% achieve functional independence (modified Rankin scale 0-2), meaning ability to manage self-care and employment. Advanced age and large hemorrhage volume predict poor prognosis; younger patients and smaller bleeds recover more favorably.
Prevention and Risk Reduction: Managing Future Hemorrhage Risk
Once a patient survives hemorrhage, recurrence prevention becomes paramount. Hypertension management remains cornerstone, target blood pressure <140/90 mmHg reduces hemorrhage recurrence by approximately 20-25%. Compliance challenges are real; many patients struggle with daily medication adherence despite understanding risks.
Anticoagulation decisions require nuanced discussion. Patients with atrial fibrillation or thrombotic history needing anticoagulation post-hemorrhage face genuine dilemma: bleeding risk versus clot risk. Some resume anticoagulation after 1-2 weeks if hemorrhage wasn't massive and reversal agents prevented expansion; others defer indefinitely. Individual risk stratification using CHA2DS2-VASc (stroke risk in AFib) versus HAS-BLED (bleeding risk) scores guides discussions, though no validated algorithm perfectly predicts optimal timing.
AVM hemorrhage survivors have 1-2% annual rebleeding risk; annual risk increases modestly after prior hemorrhage. Stereotactic radiosurgery or surgical resection can eliminate this risk but carries operative morbidity. Conservative management with seizure prophylaxis and follow-up imaging suits small AVMs in eloquent brain regions, whereas surgically accessible lesions in non-critical areas usually warrant resection or radiosurgery.
Neurological follow-up imaging at 3, 6, and 12 months documents recovery progression and detects delayed complications (hydrocephalus, porencephaly, fluid-filled cavity replacing hemorrhage).