Genital warts and condyloma trigger anxiety in many people, and yet, they are among the most common sexually transmitted infections worldwide. According to the World Health Organization (WHO), almost all sexually active people will contract human papillomavirus (HPV) at some point in their lives, making it one of the most common sexually transmitted infections worldwide.
The reassuring reality? These infections are treatable. A broad spectrum of evidence-based therapies exists, from cryotherapy and topical agents to CO₂ laser ablation and surgical excision, each tailored to the nature, location, and extent of lesions. In Turkey, specialized medical teams manage these conditions with discretion, clinical expertise, and internationally recognized standards of care.
This guide offers an in-depth, medically accurate overview of genital warts and condyloma: what they are, how they spread, how they are diagnosed, and, most importantly, what treatment options are available for both men and women. Consider it the kind of clear, honest explanation you deserve from a trusted physician.
What are genital warts and condyloma?
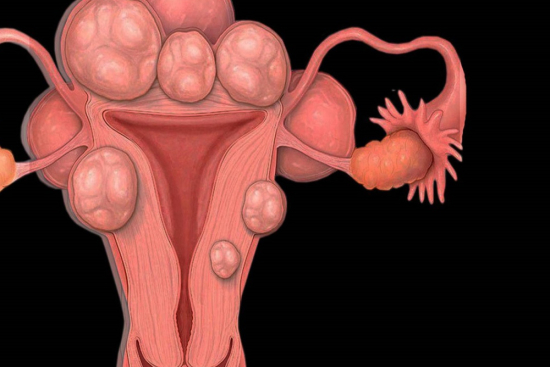
Genital warts, medically termed condylomata acuminata, are benign epithelial growths that develop on the skin and mucous membranes of the genital, perineal, and perianal regions. They are caused by specific low-risk genotypes of the human papillomavirus (HPV), most commonly HPV types 6 and 11, which account for approximately 90% of all visible genital wart cases according to the Centers for Disease Control and Prevention (CDC).
Some HPV strains are considered high-risk because they are linked to cancers of the cervix, vulva, vagina, penis, anus, and oropharynx. A visible genital wart does not inherently indicate cancer risk; however, co-infection with high-risk HPV strains is possible, which underscores the necessity of comprehensive medical evaluation whenever condyloma is identified.
Symptoms of genital warts
Genital warts can present across a spectrum of morphologies, which is why visual self-diagnosis is unreliable and professional assessment is always recommended. The main presentations include:
- Soft, flesh-colored or slightly pink papules, often multiple, flat or slightly raised, grouped in clusters;
- Filiform or cauliflower-like projections (classic condylomata acuminata), sometimes described as "cock's combs";
- Keratinized, harder lesions resembling common skin warts, more often seen in immunocompromised individuals;
- Lesions ranging from a few millimeters to several centimeters in diameter when untreated and confluent.
Importantly, the histological architecture of male and female lesions is identical. The primary distinction lies in anatomical location, not in the nature of the infection itself.
Where genital warts appear in men and women
In women, genital warts most commonly appear on the vulva, vaginal walls, cervix, or around the anus. In men, they are usually found on the penis, foreskin, scrotum, or perianal area. In both sexes, lesions may also develop inside the anal canal, especially after anal sexual contact.
How are genital warts and condyloma transmitted?
HPV is transmitted primarily through direct skin-to-skin or mucosa-to-mucosa contact with an infected individual, regardless of whether visible lesions are present. This is a critical nuance: a person can carry and transmit HPV without ever developing recognizable warts. The incubation period, from initial infection to visible lesion appearance, ranges typically from three weeks to eight months, though latent infection can persist for years.
Established transmission routes include:
- Vaginal, anal, or orogential sexual intercourse with an HPV-positive partner;
- Skin-to-skin genital contact without penetration (condoms do not fully protect against this route);
- Sharing of non-sterilized sex toys between partners;
- Vertical transmission from mother to newborn during vaginal delivery, a rare but documented cause of recurrent respiratory papillomatosis in neonates (HPV types 6 and 11).
Consistent condom use significantly reduces, though does not eliminate, transmission risk, as HPV can infect areas of the genitals not covered by a condom. For this reason, vaccination remains the most effective preventive strategy.
How genital warts are diagnosed ?
Early and accurate diagnosis is fundamental to limiting disease progression, minimizing transmission, and ruling out co-existing pathology. The diagnostic workup is multifaceted and adapted to the patient's sex, symptoms, and risk profile.
Visual and clinical examination
The first step is a thorough visual inspection of the genital, perineal, and perianal regions by a qualified specialist, dermatologist, gynecologist, urologist, or proctologist depending on the presentation. Application of 3–5% acetic acid (acetowhitening test) may help highlight subclinical lesions that blanch on contact, although this test has limited specificity and should not be used as a standalone diagnostic criterion.
Complementary examinations
- Anoscopy: indicated when perianal or intra-anal lesions are identified or suspected; allows direct visualization of the anal canal and lower rectum;
- Urethroscopy: recommended for men when condyloma is present at or near the urethral meatus, to assess intra-urethral extension;
- Colposcopy and cervical cytology (Pap smear / liquid-based cytology): mandatory for women with confirmed condyloma, to evaluate the cervix for HPV-related dysplasia (CIN) or co-existing high-risk HPV infection;
- HPV genotyping (PCR-based): can identify the specific HPV types present and distinguish low-risk from high-risk strains, though not routinely required in all cases;
- Biopsy with histopathological analysis: indicated for atypical, pigmented, ulcerated, or treatment-resistant lesions to exclude squamous intraepithelial neoplasia or invasive carcinoma.
Partner notification and screening
Clinical guidelines consistently recommend informing recent sexual partners so that they can undergo appropriate examination and, where indicated, treatment. This step is not only ethically important, it is epidemiologically essential to interrupt the transmission chain.
Prevention: HPV vaccination and protective strategies
Prevention of HPV infection relies on a combination of behavioral measures, barrier contraception, and, most effectively, prophylactic vaccination.
HPV vaccine and prevention
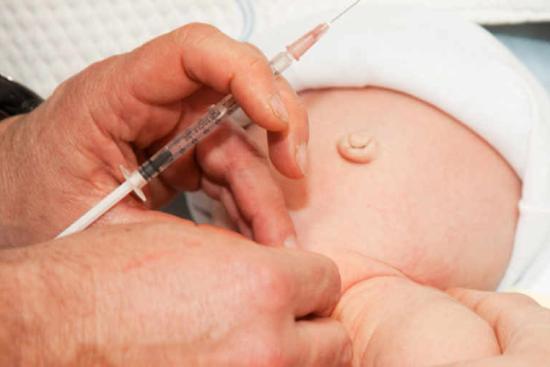
Three HPV vaccines have been developed and approved by regulatory agencies globally: Cervarix® (bivalent, HPV 16/18), Gardasil® 4 (quadrivalent, HPV 6/11/16/18), and Gardasil® 9 (9-valent, covering HPV 6, 11, 16, 18, 31, 33, 45, 52, and 58). Gardasil® 9 is now considered the standard of care in most international guidelines.
Clinical trial data and post-marketing surveillance have demonstrated that Gardasil® 9 prevents over 90% of HPV-attributable cancers and approximately 90% of genital warts. The vaccine is most effective when administered prior to sexual debut, ideally between ages 9 and 14. However, catch-up vaccination is recommended up to age 26 for all individuals, and may be considered up to age 45 following shared clinical decision-making.
Critically, vaccination does not treat existing HPV infections or condyloma, it prevents future infection with the targeted genotypes. Individuals who are already sexually active may still benefit from partial protection against strains they have not yet encountered.
Additional preventive measures
- Consistent and correct use of male or female condoms, reduces HPV transmission risk by approximately 70% when used consistently (Winer et al., NEJM, 2006);
- Mutual monogamy with a confirmed HPV-negative partner;
- Regular STI screening, at minimum annually for sexually active individuals;
- Avoidance of tobacco use, smoking is an independent risk factor for HPV persistence and associated dysplasia progression.
Treatment options for genital warts in Turkey: A clinical overview
No currently available therapy can eradicate HPV from the body. What modern treatments achieve is the clearance of visible lesions, alleviation of symptoms, and reduction of the viral load at the infection site, thereby decreasing (though not eliminating) transmission risk. Spontaneous regression of genital warts occurs in approximately 30% of cases within 4 months, and in up to 90% within 2 years in immunocompetent individuals, a fact worth communicating to patients during shared decision-making.
Treatment selection depends on several factors: lesion morphology, size, number, and location; patient preference; available resources; presence of immunosuppression; and pregnancy status. No single treatment has been shown to be universally superior to others, and combination approaches are often used for extensive or recalcitrant disease.
| Treatment | Setting | Clearance rate* | Best suited for |
| Imiquimod 5% | Home / clinic | 50–75% | External warts, small to moderate |
| Podophyllotoxin 0.5% | Home | 45–77% | External keratinized lesions |
| TCA 80–90% | Clinic only | 60–80% | Mucosal / moist lesions |
| Cryotherapy | Clinic | 63–88% | Small, few lesions; any site |
| CO₂ Laser | Surgical / OR | ~90% | Extensive, recurrent, or internal lesions |
| Surgical excision | Surgical / OR | ~90% | Pedunculated or large single warts |
| Electrosurgery | Clinic / OR | ~94% | Multiple warts, broad areas |
* Clearance rates refer to complete resolution of treated lesions at end of therapy; recurrence rates vary independently. Data compiled from CDC 2021 STI Treatment Guidelines and European guidelines (IUSTI 2012).
Patient-applied topical therapies
These agents are prescribed for self-application by the patient on external, easily accessible lesions. They are generally not appropriate for internal (vaginal, anal, cervical) locations.
- Imiquimod (3.75% or 5% cream): helps the immune system fight HPV and gradually remove genital warts. It is usually applied three times a week for several weeks. Mild redness, irritation, or burning sensations may occur during treatment. This medication is generally not recommended during pregnancy.
- Podophyllotoxin (0.5% solution or 0.15% cream): works by destroying wart tissue directly. It is applied at home in short treatment cycles over several weeks. It can be effective for small external genital warts but must never be used during pregnancy.
- Sinecatechins (15% ointment): a plant-based treatment derived from green tea extract that helps the body clear external genital warts. It is applied several times daily and is mainly recommended for patients with a healthy immune system. It is not suitable for internal warts.
In-clinic treatments for genital warts
-
Trichloroacetic acid (TCA, 80–90%): Applied carefully by a physician directly onto wart tissue, causing immediate protein coagulation and chemical necrosis. Particularly effective on moist mucosal surfaces (vaginal, anal, urethral). Sessions are repeated weekly until resolution. Suitable during pregnancy. Side effects include local burning and occasional ulceration if over-applied.
Cryotherapy for genital warts
Cryotherapy involves the controlled application of liquid nitrogen (-196°C) via a cryoprobe or spray, inducing intracellular ice crystal formation and cellular destruction. Sessions last 10–30 seconds per lesion and are typically repeated every 1–3 weeks. Complete clearance is achieved in 63–88% of patients over multiple sessions; however, recurrence rates of 25–40% have been reported in longer-term follow-up.
Cryotherapy is generally well tolerated, requires no anesthesia for external lesions, and does not produce scarring in most cases. It is considered safe in pregnancy. Side effects include transient pain, blistering, and depigmentation in darker skin types.
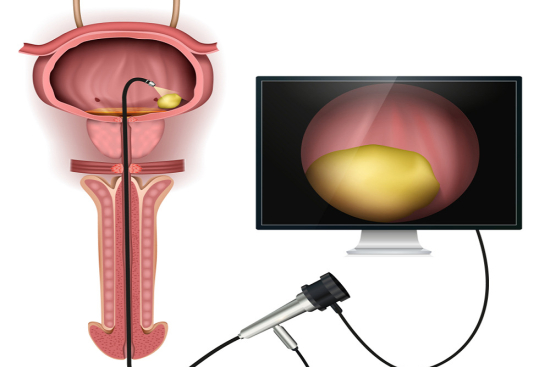
CO₂ laser treatment for genital warts
CO₂ laser therapy is widely regarded as the treatment of choice for extensive, multifocal, recurrent, or anatomically challenging condyloma, including intra-vaginal, intra-urethral, and intra-anal lesions. The laser vaporizes infected tissue with exceptional precision, achieving complete clearance in approximately 90% of cases in experienced hands.
Clinical advantages of CO₂ laser ablation include:
- High precision with minimal thermal damage to surrounding healthy tissue;
- Simultaneous coagulation of feeding blood vessels, reducing intraoperative bleeding;
- Access to lesions inaccessible to cryotherapy or topical agents;
- Lower recurrence rates compared to chemical destruction in properly selected patients;
- Healing typically complete within 2–4 weeks with excellent cosmetic outcome;
- Performed under local or general anesthesia depending on lesion extent, ambulatory procedure in most cases.
Important nuance: while laser ablation achieves superior local lesion clearance, it does not eradicate HPV from surrounding tissue. Studies have reported recurrence rates of 20–30% at 6 months, emphasizing the importance of follow-up and, in some cases, adjuvant topical immunotherapy.
The partner clinics of TurquieSanté are equipped with the latest generation medical CO₂ laser systems and staffed by dermatosurgeons and urologists trained to international standards, including JCI-accredited facilities.
Surgical excision
For pedunculated, large, or anatomically localized condyloma, sharp excision using scissors or a scalpel remains a fast and highly effective approach. Complete clearance rates approach 90% in a single session. A major advantage is the availability of the excised tissue for histopathological analysis, essential whenever an atypical or pigmented lesion raises concern for dysplasia or malignancy.
Electrosurgery (Electrodesiccation and Curettage / Diathermy)
High-frequency electrical current is used to destroy and coagulate wart tissue. This technique offers high short-term clearance rates and is particularly effective for multiple or widespread genital warts. It is often combined with curettage and is usually performed under local anesthesia. Because the procedure generates surgical smoke, appropriate protective measures and ventilation are used during treatment.
Do genital warts increase cancer risk?
One of the most common sources of patient anxiety is the perceived link between genital warts and cancer. This concern deserves a carefully nuanced answer, neither dismissive nor alarmist.
Condylomata acuminata,the visible warts caused by HPV 6 and 11, are classified as benign lesions and are NOT considered premalignant in themselves. Low-risk HPV strains that cause genital warts are generally not linked to cancer. Current evidence does not support the notion that treating genital warts prevents future cancer.
However, the following important nuances apply:
- Concurrent infection with high-risk HPV types (16, 18, 31, 33, 45) is biologically independent of low-risk HPV infection but can occur simultaneously in the same individual;
- Women diagnosed with condyloma should always receive a cervical cytology and, where indicated, HPV genotyping and colposcopy, to screen for cervical intraepithelial neoplasia (CIN) caused by independent high-risk HPV infection;
- In immunocompromised patients (HIV-positive individuals, transplant recipients, those on immunosuppressive therapy), low-risk HPV lesions may behave more aggressively and transformation to verrucous carcinoma, though rare, has been documented;
- Buschke-Löwenstein tumors, giant condylomata, represent a locally invasive variant associated with HPV 6 and 11 that can cause significant tissue destruction without true metastatic potential.
In summary: a diagnosis of genital warts should not cause fear of cancer, but it should prompt thorough evaluation to rule out co-existing high-risk HPV infection, particularly in women, where cervical cancer prevention is a critical public health priority.
Can genital warts come back after treatment?
Recurrence is one of the most clinically challenging aspects of condyloma management. Population studies estimate that 20–30% of patients experience recurrence of visible lesions within 3–6 months of completing treatment. This does not necessarily reflect treatment failure, it reflects the persistent latency of HPV in the basal epithelium of apparently normal skin surrounding treated lesions.
Factors associated with higher recurrence rates include:
- Immunosuppression (HIV infection, organ transplantation, long-term corticosteroid use);
- Active tobacco smoking, independently associated with impaired local mucosal immunity and HPV persistence;
- Untreated or unscreened sexual partners, representing a vector for reinfection;
- Extensive initial lesion burden at diagnosis;
- Incomplete treatment cycles or premature discontinuation of therapy.
Strategies to minimize recurrence include treating all sexual partners, optimizing immune function (smoking cessation, HIV treatment optimization), completing full treatment courses, and ensuring structured follow-up at 3, 6, and 12 months post-treatment. In cases of true recalcitrant condyloma, intralesional interferon alpha injections or systemic immunomodulatory approaches may be considered by a specialist.
Why choose Turkey for genital wart treatment?
Turkey has emerged as a leading international destination for medical care across dermatology, urology, and gynecology, a positioning backed not merely by competitive pricing, but by measurable quality benchmarks.
- Internationally trained specialists: Turkish dermatosurgeons, urologists, and gynecologists frequently receive postgraduate training in European and American academic medical centers, and remain engaged with current international literature and treatment protocols;
- Advanced technological infrastructure: state-of-the-art CO₂ laser platforms, high-definition endoscopy suites for anoscopy and urethroscopy, and colposcopy equipment meeting EU standards;
- JCI accreditation: partner clinics of TurquieSanté have obtained Joint Commission International accreditation, the global benchmark for patient safety and quality of care;
- Comprehensive, discreet care pathways: from initial teleconsultation to post-treatment follow-up, multilingual care coordinators ensure seamless, confidential management;
- Cost efficiency: treatment costs in Turkey are typically 40–70% lower than equivalent care in Western Europe, without compromise in clinical standard.
Turquie Santé connects patients with certified specialists in JCI-accredited institutions, ensuring that every stage of care, from diagnosis through treatment and follow-up, meets the highest international standards.